Seeking investment
biogelx is a seeking funding to commercialise UNI-GEL –
a synthetic peptide hydrogel, that enables in-vivo modelling in-vitro.
Technology
IP protected
Sector
Drug development / Regenerative medicine
£2 million
Target raise
Our mission is to be the gel standard of choice for global drug development and regenerative medicine.
Biogelx helps organizations working in translational science, drug discovery and tissue engineering to “model in vivo conditions in vitro”. We do this through designing, manufacturing and supplying synthetic hydrogels for 3D cell culture and bioprinting. Our products are reproducible and scalable, meaning they are suitable for fundamental research right through to pharmaceutical decision making.
- No batch-to-batch variation, consistency is key.
- Hydrogel technology with the broadest stiffness range (~0.5 to 100kPa).
- Functionalised with biomimetic sequences to support good cell attachment & growth.
- Formulations can be optimised for 3D bioprinting applications.
- Gelation does not require temperature or pH adjustment, just add water and media.
How UNI-GEL works
UNI-GEL is a platform solution for drug discovery and regenerative medicine through the ability to
- mimic the human body to support cell culture for drug discovery.
- flex to the required tunability to mimic different tissues of the body
- be used within printable biocompatible bio-inks for regenerative medicine.
Target markets
- Short-comings in the use of the market standard (Matrigel) are impeding effective drug discovery and regenerative medicine techniques.
- Regenerative medicine requires biomaterials, scaffolds and cells generated from robust and reliable materials
- 3D-Bioprinting offers potential to implant tissue therapeutics rather than whole organs in a consistent and safe manner
- Cell Based Assay Mkt $22bn in 2025
- Regenerative Market $18bn in 2025
B2B Proposition:
1.HYDROGELS that are a superior alternative to Matrigel
2.BIO-INKS which will be utilized in large scale regenerative medicine.
Peptide hydrogels combine all the key attributes that are essential for robust and highly reproducible cell-derived data.
Ease of use and regenerative adaptability make biogelx a future market leader.
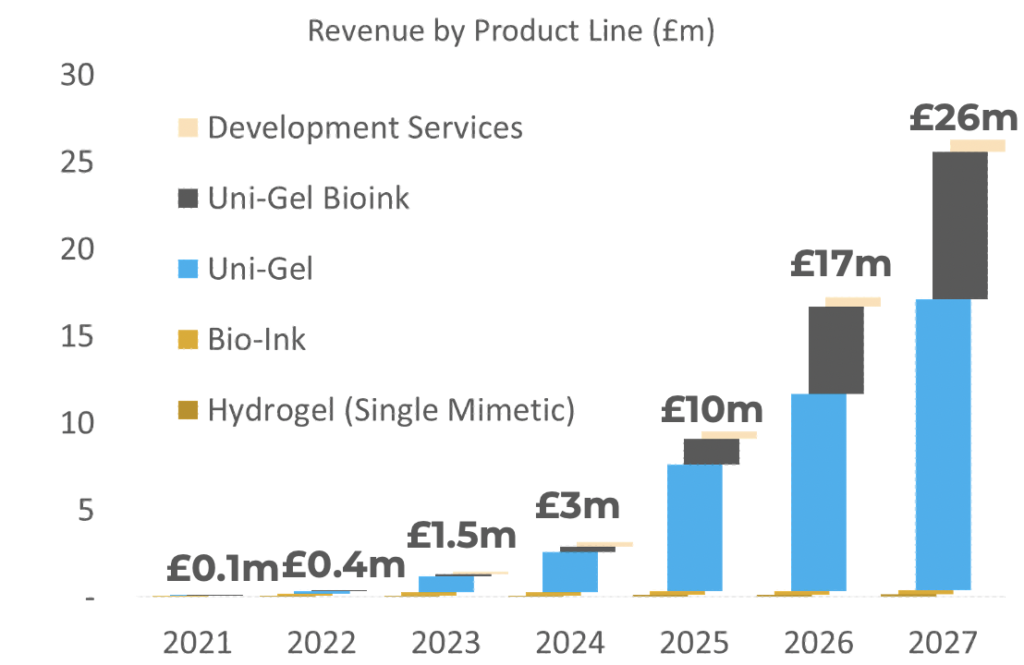
Financial projections
Live client portfolio
Commercial
GSK, Charles River, Eli Lilly
Distributor
Merck, Cedarlane, AFS Bio
Academic / commercial collaborations
Reprocell, Clyde Biosciences, Newcells
PhD university projects
Edinburgh, Glasgow, Manchester




To learn more about our products please watch the video below to hear Dr Chris Allan – our product development specialist – talk about our bioinks as part of a recent webinar.
| Company | BioGelx Ltd BioCity Scotland, Newhouse, Scotland |
| Traction | Achieving ISO9001 Certification Recruitment of 7 new PhD projects utilising either the hydrogels or bioinks due to start in October 2021. Establishing a Scientific Advisory Board in November 20 – Professors Matthew Dalby and Manuel Salmeron-Sanchez (Glasgow University) and Paul Smith (CEO Bioascent). Securing space to establish cell culture capabilities (April 2021) Attracting an experienced cell culture expert through UKRI Innovation Scholar secondment; starts July 2021. |
| Valuation | £5m pre-money |
| Investment sought | £2.0m for 28.6% equity (Q3 2021) |
| Use of funds | Develop Product Portfolio to include growth factors to enhance biological performance. Implement cell culture capabilities within laboratories at Biocity Glasgow Scientific team expansion 5 scientists/cell biologists and 2 business development Sales and marketing team expansion Capital expenditure to support technology development. Fund Collaborations to generate case studies and market evidence |
| Existing investors | University of Strathclyde Scottish Investment Bank Founders EIS Investors |
| Leadership | Dr Alison Clayton – CEO. 25yrs+ in operational and scientific leadership. Dr Mhairi Rodgers – Heads of Ops. Specialist in biotechnology and 3D biomaterials. Dr Chris Allan – Senior Scientist, responsible for new product development. John Waddell – Chairman. Extensive experience of driving value to successful exit. Margaret Temple – Non-Exec. Experienced BD and exiting in the Life Sciences Industry. Prof Rein Ulijn – Founder and Director, Champion of biomolecular nanoscience |
Disclaimer
The content of this communication has not been approved by an authorised person within the meaning of the Financial Services and Markets Act 2000. Reliance on this communication for the purpose of engaging in any investment activity may expose an individual to a significant risk of losing all of the property or other assets invested.


